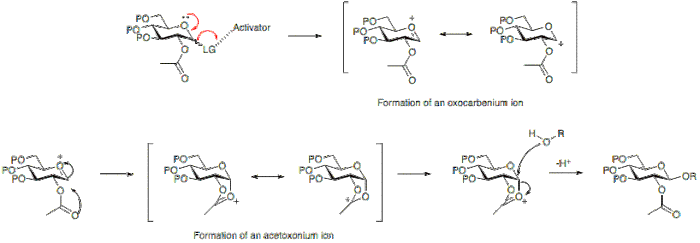
Neighbouring group participation
The stereochemical outcome of a glycosylation reaction may in certain cases be affected by the type of protecting group employed at position 2 of the glycosyl donor. A participating group, typically one with a carboxyl group present, will predominantly result in the formation of a β-glycoside. Whereas a non-particiapting group, a group usually without a carboxyl group, will often result in an α-glycoside.Below it can be seen that having an acetyl protecting group at position 2 allows for the formation for an acetoxonium ion intermediate that blocks attack to the bottom face of the ring therefore allowing for the formation of the β-glycoside predominantly.
Alternatively, the absence of a participating group at position 2, allows for either attack from the bottom or top face. Since the α-glycoside product will be favoured by the anomeric effect, the α-glycoside usually predominates.

No comments:
Post a Comment